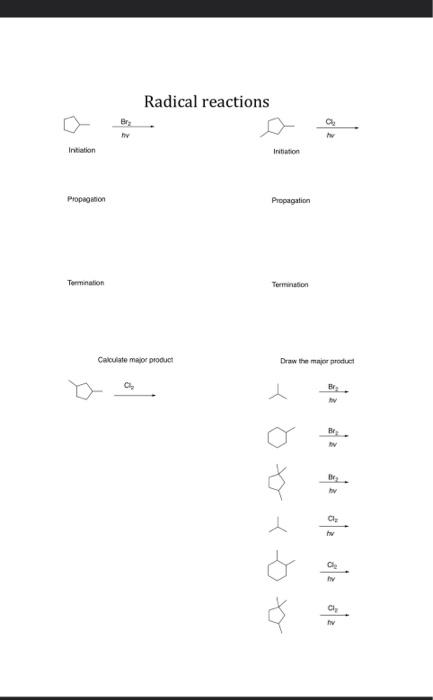
Radical reactions are a fundamental concept in organic chemistry, playing a crucial role in various chemical processes and industrial applications. These reactions involve the formation and participation of free radicals, which are highly reactive species with unpaired electrons. Understanding the mechanisms behind radical reactions is essential for students and professionals alike, as it provides insights into how these reactions proceed and how they can be controlled.
The process of radical reactions typically consists of three key steps: initiation, propagation, and termination. Initiation involves the breaking of a bond to form radicals, often requiring energy from light or heat. This step is critical because it sets the stage for the subsequent reactions. Once initiated, the radicals then participate in propagation steps, where they react with other molecules to generate new radicals, continuing the chain reaction. Finally, termination occurs when two radicals combine to form a stable molecule, effectively ending the chain reaction.
In the context of radical halogenation, such as the chlorination of ethane, these steps are clearly illustrated. The initiation phase begins with the homolytic cleavage of a σ bond, resulting in the formation of two radicals. These radicals then engage in propagation steps by abstracting hydrogen atoms from the alkane, leading to the formation of new radicals. This cycle continues until the radicals eventually terminate by combining with each other, forming stable products.
Understanding the intricacies of radical reactions not only enhances one's grasp of organic chemistry but also has practical implications in various fields, including polymer science, pharmaceuticals, and materials engineering. By delving deeper into the mechanisms of radical reactions, we can better appreciate their significance and potential applications in modern chemistry.
The importance of radical reactions extends beyond academic interest, influencing numerous industries that rely on chemical processes for product development and innovation. For instance, in the production of plastics and synthetic fibers, radical reactions are utilized to create long-chain polymers through addition mechanisms. Additionally, in the field of medicine, radical reactions play a role in drug synthesis and the development of new therapeutic agents.
Moreover, the study of radical reactions contributes to our understanding of environmental chemistry, particularly in the context of atmospheric processes and the degradation of pollutants. Free radicals are involved in the breakdown of harmful substances, making them a vital component in the study of environmental impact and sustainability. As such, mastering the concepts of radical reactions equips scientists and engineers with the knowledge needed to address contemporary challenges and develop innovative solutions.
In summary, radical reactions are a cornerstone of organic chemistry, offering valuable insights into the behavior of free radicals and their role in various chemical processes. By exploring the mechanisms of initiation, propagation, and termination, we gain a comprehensive understanding of how these reactions unfold and their broader implications across different scientific disciplines.
The Mechanisms of Radical Reactions
Radical reactions are characterized by the involvement of free radicals, which are atoms or molecules with unpaired electrons. These species are highly reactive due to their tendency to seek out other electrons to achieve a stable configuration. The mechanisms of radical reactions can be broken down into three distinct phases: initiation, propagation, and termination. Each of these steps plays a crucial role in the overall process, contributing to the dynamic nature of radical chemistry.
Initiation is the first step in a radical reaction and typically involves the breaking of a bond, resulting in the formation of two radicals. This process requires an input of energy, which can come from various sources such as light (photolysis) or heat (thermal decomposition). In the case of halogenation, for example, the initiation step often involves the homolytic cleavage of a halogen molecule, like chlorine (Cl₂), yielding two chlorine radicals (Cl•). This step is essential as it creates the reactive species necessary for the subsequent propagation steps.
Following initiation, the propagation phase begins. During this stage, the radicals generated in the initiation step react with other molecules, typically alkanes, to form new radicals. This process is crucial for sustaining the chain reaction. For instance, in the chlorination of ethane, a chlorine radical may abstract a hydrogen atom from ethane, producing a new ethyl radical (CH₃CH₂•) and hydrogen chloride (HCl). The newly formed radical can then react with another molecule of chlorine, continuing the cycle and generating more radicals. This propagation phase is responsible for the amplification of the reaction, allowing it to proceed efficiently without the need for continuous energy input.
Finally, the termination step marks the end of the radical chain reaction. This occurs when two radicals combine to form a stable molecule, thereby eliminating the reactive species. Termination can happen through various pathways, such as the combination of two radicals or the reaction of a radical with a neutral molecule. For example, two chlorine radicals might combine to form a chlorine molecule (Cl₂), while an ethyl radical could react with a chlorine molecule to produce 1-chloroethane (CH₃CH₂Cl). The termination step is vital for controlling the reaction and preventing excessive radical accumulation, which could lead to unwanted side reactions.
In conclusion, the mechanisms of radical reactions—initiation, propagation, and termination—are fundamental to understanding how these reactions proceed. Each step contributes uniquely to the overall process, highlighting the dynamic interplay between reactive species and their interactions with other molecules. By grasping these mechanisms, chemists can better predict and control radical reactions, paving the way for advancements in various scientific and industrial applications. 😊
Propagation in Radical Halogenation
Propagation is a critical phase in radical halogenation, where the initial radicals formed during the initiation step actively engage in further reactions. This step is characterized by the continuous generation of new radicals, which allows the reaction to sustain itself without the need for additional energy input. In the context of halogenation, such as the chlorination of ethane, the propagation phase is where the majority of the reaction occurs, leading to the formation of various products.
During propagation, the chlorine radicals produced in the initiation step react with the alkane, typically ethane (C₂H₆), by abstracting a hydrogen atom. This hydrogen abstraction results in the formation of a new radical, specifically an ethyl radical (CH₃CH₂•), along with the release of hydrogen chloride (HCl). The ethyl radical, being highly reactive, can then interact with another molecule of chlorine (Cl₂), initiating a new cycle of radical formation. This process is not only essential for the continuation of the reaction but also determines the selectivity and yield of the final products.
The rate-determining step in radical halogenation is often the hydrogen abstraction by the halogen radical. This step is crucial because it dictates the overall speed of the reaction and influences the types of products formed. The reactivity of the alkyl radicals formed during this process also plays a significant role. Alkyl radicals are generally more reactive than the original alkane, which means they can readily engage in further reactions with other molecules, leading to the formation of additional radicals and ultimately more complex products.
As the reaction progresses, the concentration of the products increases, which can lead to the occurrence of polychlorination. This phenomenon arises when multiple chlorine atoms are added to the alkane, resulting in a mixture of mono- and poly-chlorinated compounds. For instance, in the chlorination of ethane, the presence of excess chlorine can lead to the formation of 1,1-dichloroethane and 1,2-dichloroethane, along with higher chlorinated derivatives. This complexity highlights the importance of controlling the reaction conditions to achieve the desired product, especially when aiming for monohalogenation.
To ensure selective halogenation, it is imperative to use the alkane in excess, which helps to maintain a low concentration of the reactive radicals and minimize the likelihood of polychlorination. Monohalogenation is particularly valuable in organic synthesis, as it allows for the preparation of specific haloalkanes that can serve as intermediates in the synthesis of more complex molecules. The ability to control the reaction pathway and product distribution underscores the significance of understanding the propagation mechanism in radical halogenation.
In summary, the propagation phase in radical halogenation is pivotal for the continuation of the reaction and the formation of various products. Through the continuous generation of radicals and their subsequent reactions, the process demonstrates the dynamic nature of radical chemistry. By carefully managing the reaction conditions and understanding the underlying mechanisms, chemists can harness the power of radical reactions to achieve targeted outcomes in organic synthesis. 😊
Termination in Radical Reactions
The termination step in radical reactions marks the conclusion of the chain reaction, where the reactive radicals combine to form stable molecules. This phase is crucial for maintaining the balance of the reaction and preventing the formation of unwanted by-products. In the context of radical halogenation, the termination step ensures that the reaction does not continue indefinitely, thus controlling the extent of the reaction and the types of products formed.
During termination, two radicals can combine to form a stable molecule, effectively ending the chain reaction. This process can occur through various mechanisms, such as the direct combination of two radicals or the reaction of a radical with a neutral molecule. For example, in the chlorination of ethane, two chlorine radicals (Cl•) can react to form a chlorine molecule (Cl₂), while an ethyl radical (CH₃CH₂•) might combine with a chlorine molecule to produce 1-chloroethane (CH₃CH₂Cl). These combinations result in the formation of stable, non-reactive species, which are no longer capable of participating in further reactions.
The termination step is not limited to specific radicals; any two radicals can react to form a stable molecule, making this phase highly versatile. However, the probability of termination occurring depends on the concentration of radicals relative to the concentration of neutral molecules. Typically, the concentration of radicals is much lower, which means that they do not have a high chance of colliding with each other before reacting with other molecules. This dynamic leads to the propagation of the reaction for thousands of cycles before termination occurs, emphasizing the efficiency of radical reactions.
Understanding the termination step is essential for controlling the outcome of radical reactions. By manipulating the reaction conditions, such as temperature and the presence of inhibitors, chemists can influence the likelihood of termination. For instance, adding a small amount of a substance that can react with radicals can help to terminate the reaction prematurely, thus limiting the formation of undesired products. This level of control is vital in industrial settings, where the goal is to produce specific compounds with high purity and yield.
In summary, the termination step in radical reactions is a critical phase that ensures the stability of the reaction system. By allowing radicals to combine and form stable molecules, this step not only concludes the chain reaction but also plays a significant role in determining the final products. Mastery of this concept enables chemists to effectively manage radical reactions, optimizing their outcomes for various applications in both academic and industrial contexts. 😊
Selectivity in Radical Halogenation
Selectivity in radical halogenation refers to the preference of a reaction to form specific products over others, which is influenced by several factors, including the structure of the alkane and the nature of the halogen used. This selectivity is particularly important in the synthesis of haloalkanes, as it allows for the targeted formation of desired compounds. Understanding the principles behind selectivity can significantly enhance the efficiency and effectiveness of radical halogenation reactions.
One of the primary factors affecting selectivity in radical halogenation is the stability of the radicals formed during the reaction. Alkyl radicals exhibit varying degrees of stability depending on their structure. For instance, tertiary radicals are generally more stable than secondary or primary radicals due to hyperconjugation and inductive effects. This increased stability makes tertiary radicals more likely to form, leading to the preferential formation of products derived from the more stable radicals. Consequently, when a reaction involves the abstraction of a hydrogen atom from a branched alkane, the formation of a tertiary radical can result in the production of a more substituted product.
Another critical factor is the reactivity of the halogen. Different halogens have varying abilities to initiate and propagate radical reactions. For example, bromine is less reactive than chlorine, which can lead to a higher degree of selectivity in the reaction. This is because bromine tends to abstract hydrogen atoms from more substituted positions, favoring the formation of more stable radicals. In contrast, chlorine is more reactive and can abstract hydrogen atoms from less substituted positions, leading to a broader range of products. Therefore, the choice of halogen can significantly influence the selectivity of the reaction.
Additionally, the presence of substituents on the alkane can affect the selectivity of radical halogenation. Electron-donating groups can increase the stability of the resulting radicals, promoting the formation of more substituted products. Conversely, electron-withdrawing groups can decrease the stability of radicals, potentially leading to the formation of less substituted products. This interplay between substituents and radical stability highlights the importance of molecular structure in determining the outcome of radical reactions.
Moreover, the reaction conditions, such as temperature and solvent, can also influence selectivity. Higher temperatures may favor the formation of more stable radicals, while certain solvents can stabilize or destabilize radicals, thereby affecting the reaction pathway. By carefully controlling these parameters, chemists can manipulate the selectivity of radical halogenation reactions to achieve the desired products.
In conclusion, selectivity in radical halogenation is a multifaceted concept influenced by the stability of radicals, the nature of the halogen, the structure of the alkane, and the reaction conditions. A thorough understanding of these factors enables chemists to design and execute reactions that yield specific products with high efficiency, making radical halogenation a powerful tool in organic synthesis. 😊
Stability of Radicals
The stability of radicals is a critical factor in determining the course and outcome of radical reactions. Radicals, being species with unpaired electrons, exhibit varying degrees of stability based on their structure and the surrounding environment. Understanding the factors that contribute to radical stability is essential for predicting reaction pathways and controlling the selectivity of radical reactions.
One of the primary determinants of radical stability is the degree of substitution around the carbon atom bearing the unpaired electron. Tertiary radicals, which are formed when the unpaired electron is located on a carbon atom bonded to three other carbon atoms, are generally more stable than secondary or primary radicals. This increased stability is attributed to hyperconjugation and inductive effects, which help to delocalize the unpaired electron and reduce its reactivity. For example, in the chlorination of 2-methylpropane (isobutane), the formation of a tertiary radical leads to the preferential production of 2-chloro-2-methylpropane, showcasing the influence of radical stability on reaction outcomes.
In addition to the degree of substitution, the presence of adjacent electron-donating groups can significantly enhance radical stability. These groups, such as alkyl groups, can donate electron density through inductive effects, stabilizing the radical by reducing the electron deficiency of the unpaired electron. This stabilization is particularly evident in the case of allylic and benzylic radicals, which are stabilized by resonance. Allylic radicals, for instance, can delocalize the unpaired electron over multiple carbon atoms, increasing their stability and making them more likely to form during a reaction.
Furthermore, the size of the molecule can also influence radical stability. Larger molecules tend to provide more opportunities for the unpaired electron to be stabilized through dispersion effects, which can reduce the overall energy of the radical. This principle is observed in the radical reactions of larger hydrocarbons, where the increased number of possible conformations allows for greater stabilization of the radical intermediate.
The stability of radicals is not solely dependent on the molecular structure; environmental factors such as temperature and solvent can also play a role. Higher temperatures can increase the kinetic energy of the molecules, facilitating the formation of more stable radicals. In contrast, certain solvents may stabilize or destabilize radicals, depending on their polarity and ability to interact with the radical species.
In summary, the stability of radicals is a multifaceted aspect of radical chemistry, influenced by the degree of substitution, the presence of electron-donating groups, molecular size, and environmental conditions. A deep understanding of these factors enables chemists to predict and control the outcomes of radical reactions, ensuring the efficient synthesis of desired products. This knowledge is invaluable in the fields of organic chemistry and industrial applications, where precise control over reaction pathways is essential for achieving optimal results. 😊
Resonance Structures of Radicals
Resonance structures play a significant role in the stability and behavior of radicals, particularly in organic chemistry. When a radical is formed, it can sometimes be stabilized through resonance, which allows the unpaired electron to be delocalized over multiple atoms. This delocalization not only enhances the stability of the radical but also influences its reactivity and the types of reactions it can undergo.
For example, in the case of allylic radicals, the unpaired electron can be delocalized over a conjugated system of double bonds. This resonance stabilization results in a more stable radical compared to a simple primary or secondary radical. The ability to resonate allows the radical to distribute the electron density, reducing its overall energy and making it less reactive. This is particularly evident in the radical reactions of alkenes, where the formation of a resonance-stabilized allylic radical can lead to the preferential formation of specific products.
Similarly, in aromatic systems, the presence of a radical can be stabilized through resonance with the π-electron system of the ring. This is seen in the formation of benzylic radicals, where the unpaired electron can be delocalized into the aromatic ring, significantly increasing the stability of the radical. This resonance effect is crucial in the mechanism of radical reactions involving aromatic compounds, as it allows for the formation of more stable intermediates, which can then participate in further reactions.
The concept of resonance structures is also applicable to the formation of carbocations, where the positive charge can be delocalized through resonance. While this is not directly related to radicals, it illustrates the broader principle that the stability of reactive species can be enhanced through electronic delocalization. This understanding is essential for predicting the outcomes of various chemical reactions and designing effective synthetic strategies.
Moreover, the study of resonance structures in radicals provides insight into the mechanisms of radical reactions, enabling chemists to anticipate how different substrates will behave under various conditions. By recognizing the potential for resonance stabilization, chemists can tailor reaction conditions to favor the formation of specific radicals, thereby influencing the overall reaction pathway and product distribution.
In conclusion, resonance structures are a vital aspect of radical chemistry, offering a means to stabilize reactive species and influence their reactivity. Understanding these structures allows for a more nuanced approach to the design and execution of radical reactions, ultimately leading to more efficient and selective chemical syntheses. 😊
Stereochemistry of Radical Halogenation
Stereochemistry plays a crucial role in radical halogenation reactions, influencing the spatial arrangement of atoms and the overall reactivity of the molecules involved. Unlike ionic reactions, which often follow specific mechanisms that dictate the stereochemical outcomes, radical reactions can exhibit unique stereochemical behaviors due to the nature of the reactive species involved.
In radical halogenation, the formation of a new carbon-halogen bond occurs through the abstraction of a hydrogen atom by a halogen radical. This process typically results in the formation of a new radical at the site of hydrogen abstraction. The stereochemistry of this reaction is determined by the orientation of the radical and the surrounding substituents. For example, in the chlorination of an alkane, the abstraction of a hydrogen atom from a chiral center can lead to the formation of a new chiral center, which may result in the formation of enantiomers or diastereomers, depending on the structure of the molecule.
The possibility of forming multiple stereoisomers is a hallmark of radical reactions, particularly when the substrate contains multiple chiral centers. This complexity arises because the radical can attack from either face of the molecule, leading to different configurations. However, the presence of neighboring groups can influence the reactivity and selectivity of the reaction, often favoring certain orientations over others. This phenomenon is known as the "stereochemical bias" and can significantly impact the overall outcome of the reaction.
Moreover, the stability of the resulting radicals can also affect the stereochemical outcomes. More stable radicals are more likely to form, which can lead to the preferential formation of specific stereoisomers. For instance, if a reaction produces a tertiary radical, the stability of this radical may lead to the formation of a particular stereoisomer over others, even if the initial hydrogen abstraction occurs from a less substituted position.
In addition to these factors, the solvent and reaction conditions can also influence the stereochemistry of radical halogenation. Polar solvents may stabilize certain transition states, leading to different stereochemical outcomes, while non-polar solvents may favor other pathways. The choice of solvent can thus be a critical factor in controlling the stereochemical aspects of the reaction.
Understanding the stereochemistry of radical halogenation is essential for predicting the outcomes of such reactions and for designing synthetic strategies that yield the desired stereoisomers. By considering the factors that influence the formation and stability of radicals, chemists can better navigate the complexities of radical reactions and achieve the desired stereochemical outcomes in their synthetic endeavors. This knowledge is invaluable in the fields of organic chemistry and pharmaceutical sciences, where the stereochemistry of a compound can significantly impact its biological activity and efficacy. 😊
Allylic Bromination by NBS
Allylic bromination is a significant reaction in organic chemistry, particularly when utilizing N-bromosuccinimide (NBS) as the brominating agent. This reaction is a prime example of a radical halogenation process, where the bromine atom is introduced at the allylic position of an alkene. The mechanism of allylic bromination involves several key steps, starting with the initiation phase, followed by propagation, and finally termination, similar to other radical reactions.
The initiation step in allylic bromination typically involves the formation of a bromine radical. This can occur through the homolytic cleavage of the NBS molecule, which is facilitated by light or heat. The bromine radical generated in this step is highly reactive and can abstract a hydrogen atom from the allylic position of an alkene. This abstraction results in the formation of a new radical at the allylic carbon, which is stabilized by resonance due to the adjacent double bond. This resonance stabilization is crucial, as it lowers the energy of the radical, making it more favorable for further reactions.
Once the allylic radical is formed, the propagation phase begins. The radical reacts with another molecule of NBS, leading to the formation of a bromine-substituted product and a new bromine radical. This cycle continues, allowing the reaction to proceed efficiently without the need for continuous energy input. The propagation steps are essential for the amplification of the reaction, as each cycle generates a new radical that can participate in further reactions.
The termination step occurs when two radicals combine to form a stable molecule. This can happen through various pathways, such as the combination of two bromine radicals or the reaction of a bromine radical with a neutral molecule. The termination phase is vital for controlling the reaction and preventing excessive radical accumulation, which could lead to unwanted side reactions.
One of the advantages of using NBS in allylic bromination is its ability to selectively introduce bromine at the allylic position, making it a valuable tool in the synthesis of various organic compounds. This selectivity is influenced by the stability of the allylic radical and the reaction conditions, which can be manipulated to favor the formation of the desired product. Furthermore, the use of NBS allows for the preparation of a wide range of allyl bromides, which are essential intermediates in the synthesis of more complex molecules.
In conclusion, allylic bromination via NBS exemplifies the intricate mechanisms of radical reactions, highlighting the importance of understanding the steps involved in such processes. By leveraging the properties of NBS and the inherent stability of allylic radicals, chemists can effectively carry out this reaction to produce valuable organic compounds with specific functionalities. This reaction not only showcases the versatility of radical chemistry but also underscores the significance of selecting appropriate reagents and conditions to achieve desired outcomes in organic synthesis. 😊
Radical Halogenation in Organic Synthesis
Radical halogenation plays a pivotal role in organic synthesis, serving as a versatile method for introducing halogen atoms into organic molecules. This technique is particularly useful for the preparation of haloalkanes, which are essential intermediates in various chemical transformations. The ability to selectively introduce halogens into specific positions of a molecule allows for the creation of diverse functional groups, facilitating the synthesis of complex organic compounds.
One of the primary advantages of radical halogenation is its applicability to a wide range of substrates, including alkanes, alkenes, and aromatic compounds. This broad reactivity is due to the unique nature of free radicals, which can interact with various types of molecules to form new carbon-halogen bonds. The reaction conditions, such as the choice of halogen and the presence of initiators, can be adjusted to control the selectivity and regiochemistry of the reaction, enabling chemists to tailor the synthesis of target molecules.
For instance, in the synthesis of alkyl halides, radical halogenation can be employed to achieve monohalogenation, where a single halogen atom is introduced into the molecule. This is particularly advantageous when the desired product requires a specific functional group, as the presence of a single halogen can influence the reactivity of the molecule in subsequent reactions. The use of appropriate catalysts, such as peroxides, can enhance the initiation of the radical process, leading to higher yields and improved selectivity.
Moreover, the application of radical halogenation extends beyond simple alkyl halides. It is also utilized in the synthesis of more complex molecules, such as those containing multiple functional groups or heterocyclic structures. By strategically choosing the reaction conditions and reagents, chemists can navigate the challenges associated with the synthesis of these compounds, ensuring the formation of the desired products with minimal side reactions.
In addition to its utility in the synthesis of haloalkanes, radical halogenation is also employed in the preparation of other organic compounds, such as alcohols, ethers, and esters. The introduction of halogens can serve as a precursor to various functional group transformations, such as nucleophilic substitutions or eliminations, which are critical in the development of pharmaceuticals and other industrially relevant chemicals.
The versatility of radical halogenation in organic synthesis is further highlighted by its adaptability to different reaction environments. Whether in the laboratory or industrial settings, the principles of radical chemistry can be harnessed to optimize reaction conditions and achieve the desired outcomes. This adaptability makes radical halogenation an indispensable tool for chemists seeking to expand the scope of organic synthesis and develop innovative chemical processes.
In summary, radical halogenation is a powerful technique in organic synthesis, offering a flexible and efficient means of introducing halogen atoms into organic molecules. Its broad applicability, combined with the ability to control reaction conditions and selectivity, makes it an essential strategy for the synthesis of a wide range of organic compounds. By leveraging the unique properties of free radicals, chemists can explore new avenues in organic chemistry, paving the way for the development of novel materials and pharmaceuticals. 😊