Resonance is a fundamental concept in organic chemistry that helps explain the behavior of molecules and their stability. It refers to the ability of electrons to delocalize within a molecule, resulting in multiple valid Lewis structures that collectively represent the actual structure of the molecule. This phenomenon is crucial for understanding the properties of many organic compounds, including their reactivity, acidity, basicity, and overall stability.
In the context of resonance, it's essential to recognize that no single Lewis structure can fully capture the true nature of a molecule. Instead, resonance structures are used to depict different arrangements of electrons while maintaining the same atomic positions. These structures are not separate entities but rather different ways of representing the same molecule. The most accurate representation of a molecule is often a hybrid of all possible resonance structures, which accounts for the distribution of electron density across the molecule.
The study of resonance is closely linked to other concepts such as hybridization, lone pairs, and the rules governing the formation of resonance structures. Understanding these relationships allows chemists to predict the behavior of molecules under various conditions. For instance, resonance can significantly influence the acidity and basicity of a compound by stabilizing or destabilizing certain structures. Additionally, resonance plays a key role in determining the stability of radicals, which are highly reactive species with unpaired electrons.
This article will delve into the intricacies of resonance, exploring its impact on molecular stability, the factors that determine the significance of resonance structures, and how resonance affects the chemical properties of organic compounds. By examining these aspects, we aim to provide a comprehensive understanding of resonance and its importance in the field of organic chemistry.
Understanding Resonance Structures
Resonance structures are a critical aspect of molecular chemistry, allowing scientists to visualize and understand the dynamic nature of electron distribution within a molecule. These structures are essentially different representations of the same molecule, where the positions of atoms remain constant, but the distribution of electrons varies. This variation arises due to the delocalization of electrons, particularly in the case of pi (π) bonds, which allow for the movement of electrons between different atoms.
To identify resonance structures, one must consider the presence of pi bonds and the possibility of electron movement within a molecule. A molecule must have at least one pi bond to exhibit resonance. This is because pi bonds are formed by the sideways overlap of p orbitals, creating a region of electron density above and below the plane of the molecule. The ability to move electrons between these regions leads to the formation of multiple resonance structures, each representing a different arrangement of the electrons.
The significance of resonance structures lies in their ability to illustrate the stability of a molecule. The more resonance structures a molecule has, the more stable it tends to be, as the energy is spread out over multiple structures. This stabilization is due to the delocalization of electrons, which reduces the overall energy of the molecule. As a result, resonance structures are not just theoretical constructs; they have real implications for the chemical behavior of molecules.
Moreover, the concept of resonance is closely tied to the idea of hybridization, which describes the mixing of atomic orbitals to form new hybrid orbitals. Hybridization influences the geometry of molecules and the types of bonds that can form. In the context of resonance, hybridization plays a role in determining the stability of different resonance structures, as the hybrid orbitals can accommodate the delocalized electrons more effectively.
In summary, resonance structures are essential for understanding the electronic behavior of molecules. They provide insights into the stability and reactivity of compounds, highlighting the dynamic nature of electron distribution. By studying resonance structures, chemists can better predict the properties and behaviors of organic molecules, leading to a deeper understanding of chemical reactions and interactions.
Factors Influencing the Significance of Resonance Structures
The significance of resonance structures is determined by several key factors that influence their contribution to the overall stability of a molecule. One of the primary factors is the number of resonance structures available. Molecules with a greater number of resonance structures tend to be more stable because the electron density is distributed across multiple forms, reducing the potential for localized charges and increasing overall stability.
Another important factor is the formal charge of the atoms involved in the resonance structures. Structures that minimize formal charges are generally more significant. Formal charge is calculated by subtracting the number of non-bonding electrons and half the number of bonding electrons from the number of valence electrons. Structures with lower formal charges are preferred, as they indicate a more favorable electron distribution.
Additionally, the presence of electronegative atoms can greatly affect the significance of resonance structures. Electronegative atoms, such as oxygen and nitrogen, can stabilize negative charges through their ability to attract electrons. Therefore, resonance structures that place negative charges on electronegative atoms are typically more significant than those that do not. This stabilization contributes to the overall stability of the molecule.
The spatial arrangement of atoms also plays a role in determining the significance of resonance structures. Structures that maintain a consistent geometry and minimize steric hindrance are more likely to contribute to the stability of the molecule. Steric hindrance occurs when atoms are too close to each other, leading to repulsion and increased energy. Thus, resonance structures that avoid such conflicts are more favorable.
Furthermore, the ability of a molecule to participate in resonance is influenced by its molecular structure. Molecules that have conjugated systems, where alternating single and double bonds allow for the delocalization of electrons, are more likely to exhibit resonance. Conjugation enhances the stability of a molecule by spreading out the electron density, making it more resistant to chemical reactions.
In conclusion, the significance of resonance structures is influenced by a combination of factors, including the number of structures, formal charges, the presence of electronegative atoms, spatial arrangement, and molecular structure. Understanding these factors enables chemists to predict the stability and reactivity of molecules, providing valuable insights into their behavior in various chemical contexts. By considering these elements, researchers can better navigate the complexities of molecular chemistry and enhance their understanding of the underlying principles that govern chemical interactions. 😊
Resonance and Its Impact on Acidity and Basicity
Resonance plays a pivotal role in determining the acidity and basicity of organic compounds. When a molecule can resonate, it allows for the delocalization of electrons, which can significantly influence the stability of the conjugate base or acid. This effect is particularly evident in the case of carboxylic acids, where the resonance stabilization of the conjugate base (the carboxylate ion) makes the acid stronger. The ability of the negative charge to be delocalized over multiple atoms increases the stability of the conjugate base, thereby enhancing the acid's strength.
For example, consider the resonance structures of a carboxylic acid. The negative charge on the oxygen atom can be delocalized through the adjacent carbonyl group, leading to a more stable conjugate base. This resonance effect is not only limited to carboxylic acids but also applies to other functional groups such as phenols and alcohols. In phenols, the resonance stabilization of the conjugate base (phenoxide ion) is achieved through the delocalization of the negative charge onto the aromatic ring, making phenols more acidic than typical alcohols.
Conversely, resonance can also affect the basicity of a molecule. Amines, for instance, can exhibit varying degrees of basicity based on their ability to donate electrons. The presence of electron-donating groups can enhance the basicity by increasing the availability of lone pairs on the nitrogen atom. However, if resonance effects stabilize the positive charge that forms upon protonation, the basicity may decrease. This delicate balance between resonance and electron donation illustrates the complexity of predicting acidity and basicity in organic chemistry.
Moreover, the position of substituents on a molecule can influence its acidity or basicity through resonance effects. Electron-withdrawing groups can decrease the acidity by stabilizing the conjugate base, while electron-donating groups can increase acidity by destabilizing the conjugate base. This relationship highlights the importance of understanding resonance in the context of molecular structure and function.
In summary, resonance is a powerful tool for understanding the acidity and basicity of organic compounds. By influencing the stability of conjugate bases and acids, resonance effects can significantly alter the chemical properties of molecules. This knowledge is essential for chemists in predicting and manipulating the behavior of organic compounds in various reactions and applications. 🧪
Identifying Molecules That Can Exhibit Resonance
Not all molecules can exhibit resonance; the ability to do so depends on specific structural characteristics. To determine whether a molecule can have resonance structures, one must consider the presence of pi bonds and the possibility of electron delocalization. Molecules that contain conjugated systems, such as alternating single and double bonds, are prime candidates for resonance. These systems allow for the movement of electrons between different atoms, resulting in multiple valid Lewis structures that represent the same molecule.
A classic example of a molecule that exhibits resonance is benzene. Benzene consists of a six-membered ring with three double bonds, which are arranged in a conjugated system. The delocalization of electrons in this system results in two major resonance structures, where the double bonds can shift positions around the ring. This resonance stabilization significantly contributes to the stability of benzene, making it less reactive compared to other unsaturated hydrocarbons.
Another example is the carbonate ion (CO3^2-), which contains a central carbon atom bonded to three oxygen atoms. The carbonate ion can resonate, allowing the double bond to shift between the different oxygen atoms. This resonance results in a more stable ion, as the negative charge is delocalized across the entire molecule. Such resonance effects are crucial in understanding the chemical behavior of polyatomic ions and their interactions in solution.
In addition to these examples, molecules containing conjugated dienes, such as 1,3-butadiene, can also exhibit resonance. The alternating single and double bonds in this molecule allow for the delocalization of electrons, leading to the formation of multiple resonance structures. This resonance effect is vital in understanding the reactivity of such molecules in various chemical reactions, including electrophilic additions and Diels-Alder reactions.
It is also worth noting that the presence of lone pairs on atoms can facilitate resonance. For instance, in the case of the nitrate ion (NO3^-), the nitrogen atom has a lone pair that can participate in resonance, allowing for the delocalization of the negative charge among the three oxygen atoms. This resonance stabilization is essential for the overall stability of the nitrate ion and its behavior in aqueous solutions.
In conclusion, identifying molecules that can exhibit resonance requires an understanding of their structural features, particularly the presence of conjugated systems and the ability to delocalize electrons. Examples such as benzene, the carbonate ion, and the nitrate ion illustrate the significance of resonance in enhancing molecular stability and influencing chemical reactivity. By recognizing these characteristics, chemists can better predict the behavior of molecules and their interactions in various chemical contexts. 🔬
Rules for Drawing Resonance Structures
Drawing resonance structures involves following a set of established rules that ensure the validity and accuracy of the structures. These rules help chemists visualize the delocalization of electrons and understand the stability of different resonance forms. The first rule is to maintain the same atomic positions throughout all resonance structures. This means that the connectivity of atoms should remain unchanged, even though the distribution of electrons may vary.
Next, it is essential to ensure that the total number of valence electrons remains constant across all resonance structures. This includes both bonding and non-bonding electrons. If the number of electrons changes, the structures are not considered valid resonance forms. Additionally, the overall charge of the molecule or ion must remain the same in all resonance structures. This is crucial for maintaining the integrity of the molecule’s identity.
Another critical rule is the proper placement of double bonds and lone pairs. Resonance structures should reflect the correct number of bonds and lone pairs, ensuring that the octet rule is satisfied for all atoms. For instance, in a molecule like the carbonate ion, the double bond can shift between the oxygen atoms, but the total number of bonds and lone pairs must remain consistent. This ensures that the resonance structures accurately represent the molecule’s electronic configuration.
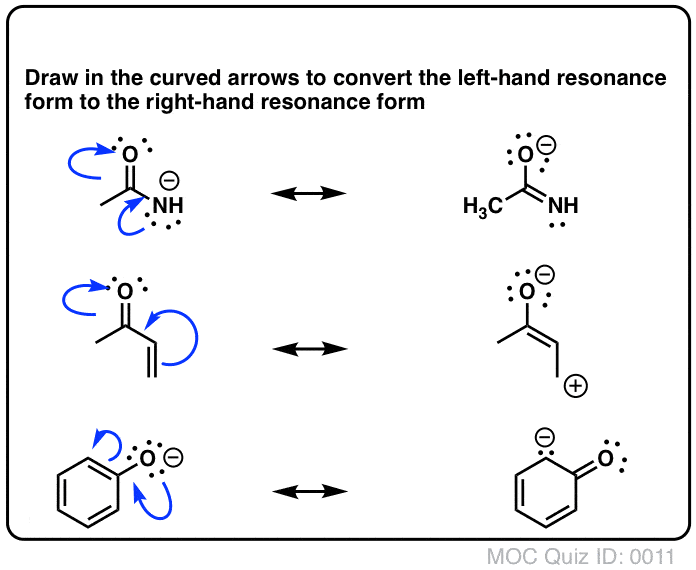
Furthermore, the use of curved arrows to indicate the movement of electrons is an essential part of drawing resonance structures. These arrows show how electrons are transferred from one position to another, illustrating the delocalization process. It is important to draw these arrows correctly, as they help in understanding the mechanism behind resonance and the stability of the resulting structures.
Lastly, the significance of each resonance structure should be evaluated based on factors such as formal charges and the stability of the resulting structures. Structures that minimize formal charges and distribute electron density evenly are generally more significant contributors to the overall resonance hybrid. This evaluation is crucial for determining which resonance structures are most relevant in understanding the behavior of the molecule.
By adhering to these rules, chemists can effectively draw and interpret resonance structures, gaining insights into the electronic behavior of molecules. This foundational knowledge is essential for predicting the properties and reactivity of organic compounds in various chemical contexts. 🧩
Major and Minor Resonance Contributors
Understanding the distinction between major and minor resonance contributors is essential for comprehending the stability and reactivity of molecules. Major resonance contributors are those that contribute significantly to the overall structure of a molecule, while minor contributors have a lesser impact. The significance of each resonance structure is determined by several factors, including the distribution of charges, the presence of electronegative atoms, and the overall stability of the molecule.
Major resonance contributors typically exhibit lower formal charges and a more favorable electron distribution. For instance, in the case of the carbonate ion (CO3^2-), the resonance structures that place the negative charge on the oxygen atoms are considered major contributors. This is because oxygen is more electronegative than carbon, allowing it to stabilize the negative charge effectively. Conversely, structures that result in higher formal charges on less electronegative atoms are deemed minor contributors, as they are less stable and less likely to occur in reality.
Additionally, the ability of a molecule to delocalize electrons plays a crucial role in determining the significance of resonance structures. Molecules that can spread out their electron density across multiple atoms are more stable and thus have more significant resonance contributors. For example, in the case of benzene, the resonance structures that allow for the delocalization of electrons around the ring are major contributors, whereas structures that do not achieve this delocalization are minor.
The stability of a resonance contributor is also influenced by the presence of conjugated systems. Molecules with conjugated systems, such as those found in aromatic compounds, can exhibit more significant resonance contributors due to the enhanced stability provided by the delocalization of electrons. This is why aromatic compounds are generally more stable and less reactive compared to their non-aromatic counterparts.
In summary, the identification of major and minor resonance contributors is vital for understanding the behavior of molecules in chemical reactions. By evaluating factors such as formal charges, electron distribution, and the presence of conjugated systems, chemists can determine which resonance structures are most relevant in describing the actual structure of a molecule. This knowledge not only aids in predicting the properties of organic compounds but also enhances the understanding of their reactivity in various chemical contexts. 🌟
The Role of Resonance in Radical Reactions
Resonance plays a crucial role in radical reactions, particularly in the stabilization of radical species. Radicals are highly reactive species with unpaired electrons, and their stability can be significantly influenced by the ability to delocalize the unpaired electron through resonance. This delocalization helps to spread out the electron density, thereby reducing the overall energy of the radical and increasing its stability.
In the case of allylic radicals, for example, the unpaired electron can be delocalized over multiple carbon atoms, leading to a more stable structure. This resonance effect is particularly evident in the allyl radical, where the unpaired electron can be shared between the terminal and the central carbon atoms. The resulting resonance structures allow for a more even distribution of the electron density, which enhances the stability of the radical. This stabilization is essential for understanding the reactivity of allylic radicals in various chemical processes, such as radical halogenation and radical polymerization.
Moreover, resonance can also influence the reactivity of radicals in different environments. For instance, in the presence of electron-donating groups, the stability of a radical can be further enhanced through resonance effects. These groups can donate electron density to the radical, helping to stabilize the unpaired electron. This is particularly important in the context of radical reactions, where the stability of the intermediate radicals can dictate the overall reaction pathway and the likelihood of certain products forming.
In addition to stabilizing radicals, resonance can also affect the mechanism of radical reactions. The ability to delocalize the unpaired electron can lead to different reaction pathways, depending on the resonance structures that are favored. This is especially relevant in the context of radical chain reactions, where the propagation steps involve the formation and consumption of radicals. The stability of the radicals formed during these steps can significantly impact the overall rate and outcome of the reaction.
In summary, resonance is a critical factor in the behavior of radicals in chemical reactions. By enabling the delocalization of unpaired electrons, resonance contributes to the stability of radical species and influences their reactivity. Understanding these effects is essential for predicting the outcomes of radical reactions and designing effective synthetic strategies in organic chemistry. 🔬
Resonance Structures of Radicals
Resonance structures of radicals are a fascinating aspect of organic chemistry, revealing how unpaired electrons can be delocalized to enhance the stability of these reactive species. Radicals, characterized by their unpaired electrons, often exhibit resonance effects that allow for the distribution of the electron density across multiple atoms. This delocalization not only stabilizes the radical but also influences its reactivity and the pathways it can take in chemical reactions.
One notable example is the allyl radical, where the unpaired electron can be delocalized over the three carbon atoms in the allyl group. This resonance leads to two major resonance structures, each depicting the unpaired electron on a different carbon atom. The ability to delocalize the electron provides the radical with increased stability, making it more likely to participate in various reactions, such as radical halogenation and radical addition reactions.
Another example is the benzyl radical, which can resonate through the aromatic ring. The unpaired electron in the benzyl radical can be delocalized across the ring, leading to resonance structures that resemble the aromatic system. This resonance effect enhances the stability of the radical, allowing it to persist longer in solution and participate in subsequent reactions. The stability conferred by resonance is crucial in understanding the mechanisms of radical reactions involving aromatic compounds.
Furthermore, the concept of resonance in radicals extends to more complex structures, such as the t-butyl radical. In this case, the unpaired electron can be delocalized through hyperconjugation, where the adjacent C-H bonds contribute to the stability of the radical. This effect is particularly significant in tertiary radicals, where the presence of multiple alkyl groups enhances the stability through increased hyperconjugation and resonance.
In conclusion, the resonance structures of radicals play a vital role in their stability and reactivity. By allowing for the delocalization of unpaired electrons, resonance effects can significantly influence the behavior of radicals in various chemical contexts. Understanding these resonance effects is essential for predicting the outcomes of radical reactions and designing effective synthetic strategies in organic chemistry. 🧪
The Importance of Lewis Structures in Organic Chemistry
Lewis structures are fundamental to the study of organic chemistry, serving as a visual representation of the bonding and electron distribution within molecules. These structures provide a clear depiction of the valence electrons, showing how atoms connect through covalent bonds and where lone pairs reside. By illustrating the arrangement of electrons, Lewis structures help chemists understand the reactivity and stability of molecules, making them an essential tool in the analysis of chemical behavior.
The construction of a Lewis structure involves several key steps. First, one must determine the total number of valence electrons available for bonding by summing the valence electrons of each atom in the molecule. Next, the atoms are connected through single bonds, with the central atom typically being the one with the lowest electronegativity. After establishing the basic framework, any remaining electrons are distributed as lone pairs on the atoms, ensuring that each atom satisfies the octet rule, which states that atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons.
However, there are exceptions to the octet rule, particularly in molecules with odd numbers of electrons or those containing elements from the third period and beyond. In such cases, the Lewis structure may include expanded octets or incomplete octets, reflecting the unique bonding capabilities of these atoms. For instance, molecules like sulfur hexafluoride (SF6) exhibit expanded octets, where sulfur can accommodate more than eight electrons due to its ability to utilize d-orbitals.
Moreover, the concept of resonance is closely related to Lewis structures, as it allows for the representation of multiple valid electron distributions within a single molecule. This is particularly useful for understanding the stability and reactivity of molecules that can exist in multiple forms. For example, the carbonate ion (CO3^2-) can be represented by several resonance structures, each showing the delocalization of the double bond among the oxygen atoms. This resonance effect not only enhances the stability of the ion but also influences its chemical properties.
In summary, Lewis structures are indispensable in organic chemistry, offering insights into the bonding and electron distribution of molecules. By following the steps to construct these structures and considering exceptions to the octet rule, chemists can better understand the behavior of organic compounds. The integration of resonance concepts further enriches the interpretation of molecular structures, providing a comprehensive foundation for analyzing chemical reactions and interactions. 🧪

